Super Refined™ excipients for parenteral drug delivery
Parenteral formulation frequently faces limitations associated with drug stability and poor solubility. Drug compatibility and the minimum concentration of drug required to produce the targeted pharmacological effect are key to excipient selection.
Your partner for parenteral formulations:
- Access specialised technical support to navigate and address your formulation challenges
- Explore our extensive offering of high purity, multi-compendial excipients listed by the FDA Inactive Ingredients Database as suitable for parenteral administration
- Our TMSuper Refined products add value to your parenteral drug formulations, reducing API degradation and aiding in successful formulation development.
Reduced API costs for injectable formulations
Our technical experts have reformulated a range of APIs with our Super Refined excipients, demonstrating a significantly higher percentage of API is retained over time when compared to formulation with standard pharmaceutical grade excipients. Reducing the degradation of your API in formulation lowers your API costs due to a decrease in the minimum efficient concentration required to produce the desired pharmacological effect over the shelf life of your product.
By reducing API degradation, less API is required per dose to reach the efficient concentration to produce the desired pharmacological effect. This not only improves the shelf life of your product but also reduces API costs.
Improving product safety profiles for injectable delivery
The removal of polar impurities reduces API/excipient interactions, minimising the presence of API degradation products.
Lengthening product shelf lives
Super Refined excipients have been demonstrated to outperform their standard compendial grade counterparts. Our excipients extend the stability of APIs over significantly longer time periods and reduce API oxidation potential – increasing drug stability.
Improving patient comfort for injectable delivery
Our Super Refined products improve patient comfort through minimising cellular irritation and allergenic reaction potential.

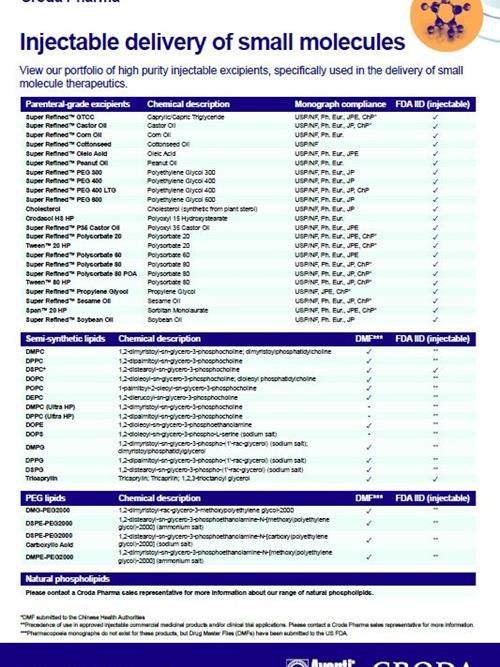
Injectable delivery of small molecules
Discover our vast portfolio of parenteral offerings used specifically in the injectable delivery of small molecules.
One category of our injectable excipient portfolio includes lipids. Lipid excipients play a pivotal role in enhancing the parenteral delivery of small molecule APIs by facilitating the formation of liposomes. Liposomes provide a stable and biocompatible carrier system for the encapsulation and solubilization of both hydrophobic and hydrophilic drug compounds. This unique capability not only improves the drug's bioavailability but also ensures a controlled and sustained release, optimizing therapeutic outcomes.
Liposomal drug delivery has shown proven efficacy in various small molecule applications, including pain management drugs as well as cancer therapies. The ability of liposomes to encapsulate drugs makes them an ideal choice for targeted delivery, addressing the specific challenges associated with these critical therapeutic areas. Our dedicated focus on developing cutting-edge lipid excipients underscores our commitment to advancing the field of injectable drug delivery, providing pharmaceutical developers with reliable solutions for formulating next-generation small molecule drugs.
Explore our injectable pharmaceutical excipients:
20 items available

Vaccine adjuvants
Our range of vaccine adjuvants can support your injectable formulation
Adjuvants are used in vaccines to accelerate, prolong, or enhance antigen-specific immune responses. Adjuvants help to raise an early, long-lasting and efficient immune response, increasing vaccine efficacy and providing a high degree of protection against the disease against which you are vaccinating.
Adjuvants enable you to reach the same level of immune protecting protection with fewer injections or a lower dose than when vaccinating with the non-adjuvanted antigen alone. (This can result in better patient compliance – less side effects/more likely to finish course if fewer vaccines are required).

Super Refined™ excipients
We have a large variety of products, ranging from various polysorbates, polyethylene glycols (PEG), esters, vegetable oils, and more.




.jpg&mn=healthcare&w=768&xr=0&yr=0&xfp=6&yfp=6&hash=024E1E255E3A56F566F39BD6B73AD51719C138999FEC74AA)
.jpg&mn=healthcare&w=768&xr=0&yr=0&xfp=6&yfp=6&hash=CC2ED77A6E51ACA367EE7726DC83D78619C138999FEC74AA)








.jpg&mn=healthcare&w=768&xr=0&yr=0&xfp=6&yfp=6&hash=763965F3AD3C9951B8AA4102D49F2C8619C138999FEC74AA)


.jpg&mn=healthcare&w=768&xr=0&yr=0&xfp=6&yfp=6&hash=8AF5F237FD30BDE03402A7A6081D2B6119C138999FEC74AA)